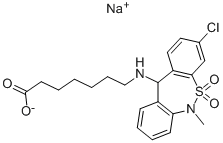
99% Tianeptine sodium salt CAS 30123-17-2
Specification
|
ITEM |
STANDARD |
RESULT |
|
Appearance |
White to yellowish powder |
White crystalline powder |
|
Solubility |
Freetly soluble In water and methanol and dichloromethane |
Conforms |
|
Identification |
By IR, To match with working standard |
Conforms |
|
(2) It gives reaction of sodium |
Conforms |
|
|
Impurities A |
≤0.1% |
Not detected |
|
Related compounds |
Any Max single impurity≤0.1% |
0.060% |
|
Total impurities≤0.4% |
0.36% |
|
|
Residual Solvents (GC) |
Acetidin≤ 0.5% |
N.D. |
|
Dichloromethane≤ 0.06% |
N.D. |
|
|
Methanol≤ 0.3% |
N.D. |
|
|
Acetone≤ 0.5% |
0.29% |
|
|
Acetonitrile≤0.041% |
0.022% |
|
|
Ethanol≤0.5% |
N.D. |
|
|
N, N-dimethylformamide≤0.088% |
N.D. |
|
|
Water |
≤ 5.0% |
3.8% |
|
Assay (HPLC) |
99.0%-101.0%(On anhydrousbasis) |
99.70% |
Application
Tianeptine sodium salt is a tricyclic antidepressant. Its antidepressant mechanism is different from that of traditional tricyclic antidepressants. It can increase the reuptake of 5-HT in the synaptic cleft. Effects of HT neuronal conduction. No affinity for choline or adrenergic receptors. The antidepressant efficacy is similar to that of tricyclics, but the tolerability is better than that of tricyclics. For the treatment of mild, moderate or severe depression, neurogenic and reactive depression, anxiety and depression with physical, especially gastrointestinal discomfort, and anxiety and depression in alcohol-dependent patients during withdrawal.